Subvisible Particle Analysis in Biopharma: Methods, Risks, and Best Practices
Subvisible particles remain one of the most important—and most difficult—quality risks in injectable pharmaceuticals and biologics. While visible particles are easier to detect and investigate, subvisible particles often occur as complex populations that can be difficult to interpret using count-only methods.
For decades, subvisible particle testing has relied heavily on Light Obscuration (LO) to report particle size and count, as described in compendial methods such as USP <787> and USP <788>. That approach remains essential for routine testing. However, today’s biologics and complex injectables are pushing the industry toward a broader expectation: in many cases, subvisible particle programs must go beyond “how many” and include particle characterization—what the particles look like, whether populations are mixed, and what they may indicate.
This shift is reflected in USP guidance such as USP <1788> and USP <1788.3>, which describe orthogonal methods and recognize the value of techniques that provide particle images and morphology.
In this guide, we cover:
-
what subvisible particles are and why they matter
-
the most common measurement methods (LO, microscopy, imaging-based techniques)
-
where USP <1788> and USP <1788.3> fit into modern best practices
-
what imaging techniques add that count-only approaches cannot
-
practical investigation best practices used by QC and development teams
Key takeaway: In modern biologics, count and size are necessary—but often not sufficient.
1) What Are Subvisible Particles—and Why Do They Matter?
Subvisible particles are typically defined as particles that are not visible to the unaided eye, but still present in the injectable product. These particles can include a wide range of materials and morphologies such as:
-
protein aggregates
-
silicone oil droplets
-
fibers
-
glass or polymer fragments
-
environmental particulates
-
process-related contaminants
Unlike visible particles, subvisible populations can be difficult to interpret because they may:
-
contain mixed particle types
-
include translucent or deformable particles
-
change over time due to settling or droplet coalescence
-
vary widely in morphology and composition
From a quality standpoint, subvisible particles matter because they may indicate:
-
formulation instability
-
manufacturing process issues
-
container closure interactions
-
handling or transport stress
-
syringe or vial-related contaminants
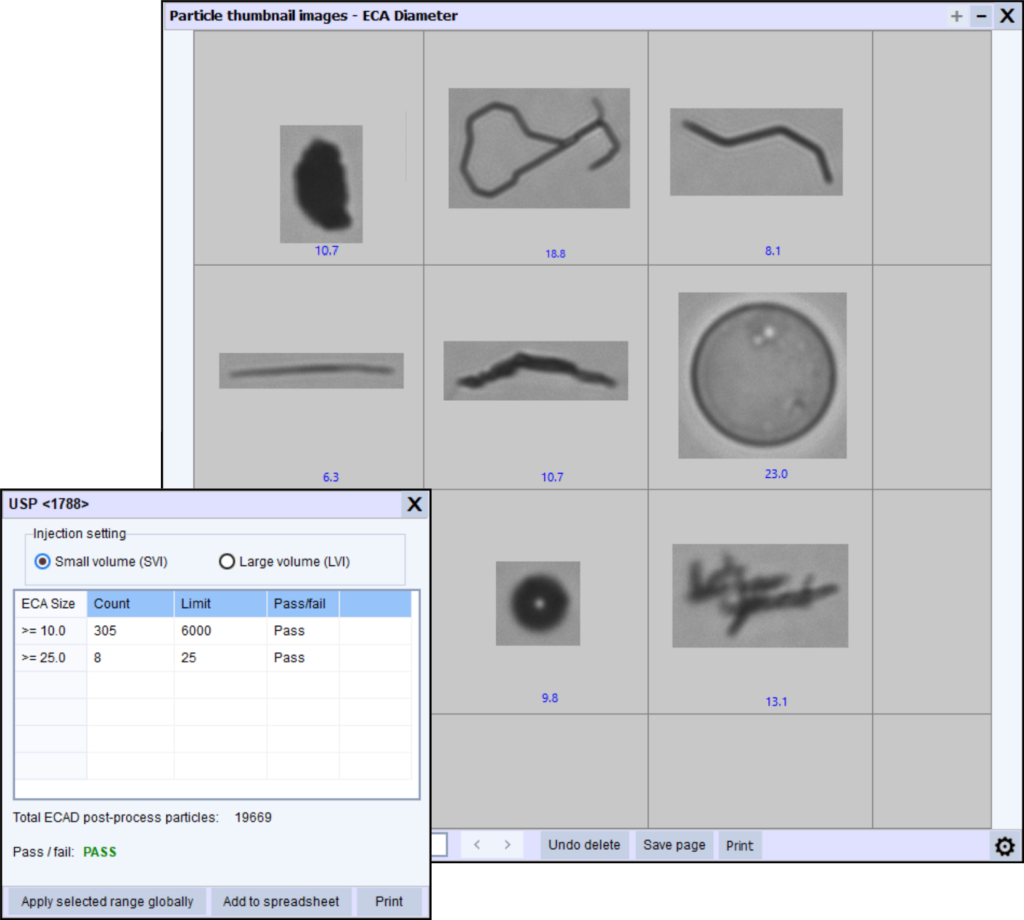
Representative particle images showing common populations such as protein aggregates, silicone oil droplets, and fibers. Image-based particle analysis helps differentiate these populations during investigations.
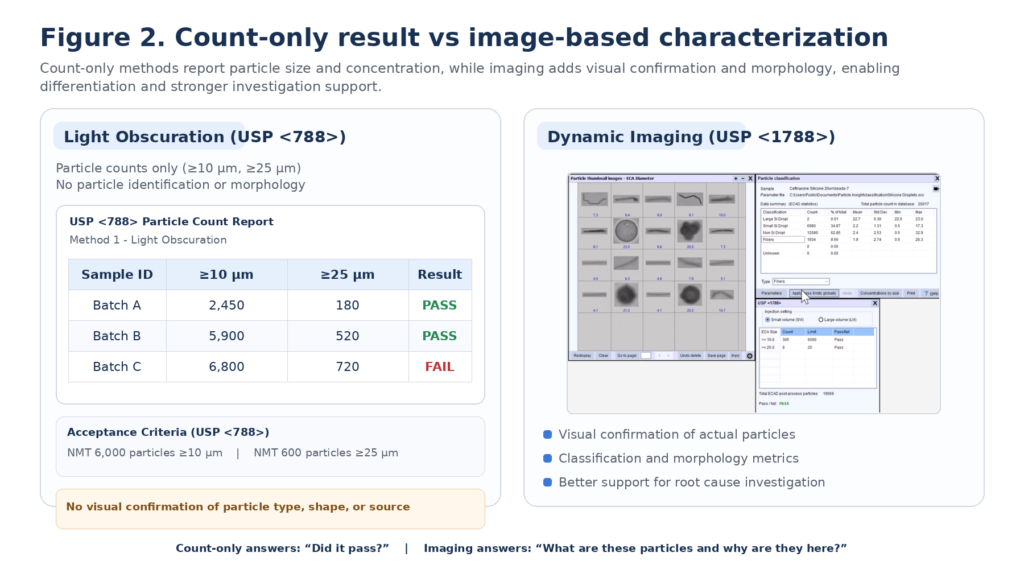
2) Why “Count-Only” Approaches Have Limitations
Light Obscuration remains the most widely used method for subvisible particle testing because it is standardized, validated, and widely accepted. It provides fast measurement of particle count and size distribution, which is essential for compendial compliance.
However, LO reports only:
-
size
-
count
…and does not provide:
-
particle identity
-
morphology
-
visual confirmation
-
differentiation of particle types
This limitation matters because many real-world subvisible contaminants—such as protein aggregates and silicone oil droplets—can be translucent, irregular, or deformable. Two very different particle populations may produce similar size/count results even though they represent very different investigation outcomes.
When the method cannot show what a particle looks like, investigations become slower and more subjective. Teams may struggle to answer basic questions such as:
-
Are these particles real or artifact?
-
Is the population uniform or mixed?
-
Are we seeing droplets, aggregates, fibers, or fragments?
-
What is the likely source?
In investigations, size and count are often the beginning—not the end—of the story.
3) Overview of Methods for Subvisible Particle Analysis
There is no single perfect method for all particle types. Different methods “see” particles differently—especially translucent, irregular, or soft particles—which is why orthogonal approaches are increasingly recommended.
Below is a practical overview of the most common methods used in biopharma.
4) Comparison Table: LO vs Microscopy vs Imaging-Based Methods
Tip: This table improves clarity for readers and helps SEO because it answers “which method should I use?” queries directly.
| Method | What it Measures | Strengths | Limitations | Best Used For |
|---|---|---|---|---|
| Light Obscuration (LO) | Size + count based on light blockage/scatter | Compendial method (USP <787>/<788>), fast, high throughput, widely validated | No images, limited particle identity, challenges with translucent/irregular particles | Routine release testing, trending, baseline monitoring |
| Membrane Microscopy | Visual inspection of filtered particles | Direct confirmation, helpful for certain contaminants | Labor-intensive, slower, operator-dependent, limited statistics | Confirmatory investigations, special studies |
| Dynamic Image Analysis (DIA) / Flow Imaging Microscopy | Images + morphology + size + count | Visual proof for every particle, morphology metrics, differentiation/classification, stronger investigation support | Typically more complex than LO; requires method setup and data interpretation | Orthogonal characterization, investigations, differentiation of particle types |
5) Light Obscuration (LO): The Compendial Foundation
Light Obscuration detects particles based on how they block or scatter light as they pass through a sensor. It remains widely used for routine QC and compendial reporting.
Strengths
-
standard method in USP <787>/<788>
-
fast counting and sizing
-
relatively high throughput
-
widely validated and accepted
Limitations
-
cannot identify particle type
-
struggles with translucent or irregular particles
-
no images or morphology
-
limited interpretability in investigations
✅ Best used for: routine release testing and trending.
6) Membrane Microscopy: Visual Confirmation with Lower Throughput
Microscopy methods typically involve filtration and manual (or semi-automated) analysis of particles.
Strengths
-
provides visual confirmation
-
can detect some particle types missed by LO
-
useful in investigations when identification is needed
Limitations
-
labor intensive
-
slower throughput
-
higher variability
-
limited statistical power unless a significant number of particles are analyzed
✅ Best used for: confirmatory investigations or targeted studies.
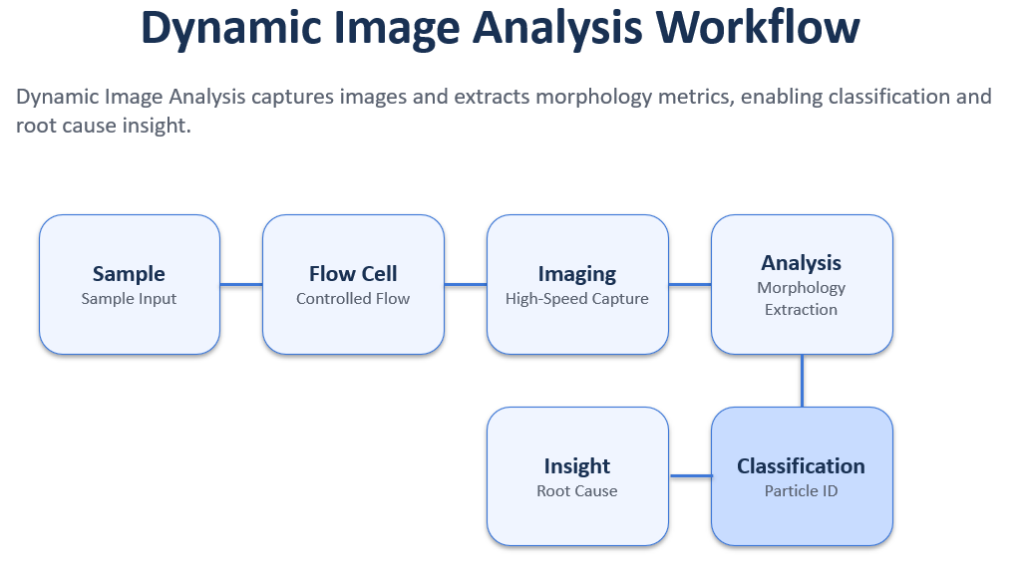
7) Dynamic Image Analysis (DIA) / Flow Imaging Microscopy: Characterization + Visual Proof
Image-based methods capture pictures of particles as they pass through a flow cell, then use software to quantify size, shape, and morphology metrics.
This method is particularly valuable because it provides:
-
images of every particle
-
morphology metrics (shape, transparency indicators, texture, etc.)
-
the ability to differentiate particle populations
-
traceable documentation for investigations
✅ Best used for: orthogonal characterization and investigation support.
Dynamic Image Analysis captures images of particles and extracts morphology metrics, enabling differentiation and stronger investigation workflows.
8) Where USP <1788> Fits: The Case for Orthogonal Characterization
USP <1788> provides a framework for subvisible particle characterization and supports the concept that:
-
different methods produce different outcomes
-
orthogonal methods improve understanding
-
characterization is essential in many situations
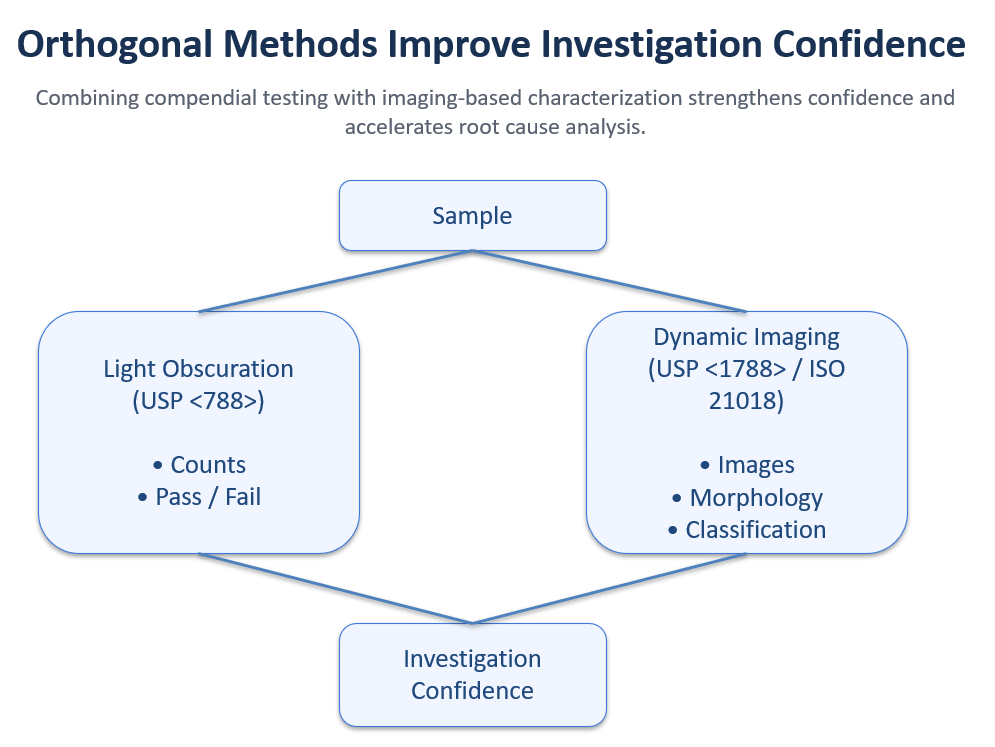
Rather than replacing compendial testing, USP <1788> supports a broader best practice:
Use multiple orthogonal approaches to improve understanding and support investigations.
This is especially relevant when:
-
particle populations are complex or mixed
-
LO results are ambiguous or unexpected
-
investigations require stronger evidence
-
the product is a complex biologic formulation
-
there is a trend or deviation requiring root cause analysis
✅ Learn more about the USP<1788> and using Dynamic Image Analysis for subvisible particle testing here
✅ Learn more about the Dynamic Image Analysis vs Micro Flow Imaging here
9) Where USP <1788.3> Fits: Imaging as a Recognized Orthogonal Method
USP <1788.3> expands guidance specifically for injectables and recognizes Dynamic Image Analysis as an orthogonal method for subvisible particle characterization.
The key principle is practical: many particle populations cannot be adequately understood through count-only results alone. Imaging-based methods provide:
-
visual confirmation
-
morphology metrics
-
differentiation of particle types
-
stronger evidence for investigations
This approach is not about replacing established LO workflows—it is about complementing them with characterization capabilities.
✅ USP <1788.3> Compliance and Subvisible Particle Characterization
Orthogonal approaches improve confidence in investigations by combining compendial testing with imaging-based characterization.
10) What Imaging Adds (and Why It Matters in QC)
Imaging-based particle analysis adds the missing layer: identity clues.
With imaging, teams gain:
-
particle images
-
size + shape data
-
morphology descriptors
-
classification possibilities
-
traceable evidence for investigations
This enables QC teams to:
-
confirm whether events are real
-
differentiate particle types
-
identify contamination sources faster
-
generate stronger deviation and CAPA documentation
-
improve confidence in quality decisions
Imaging turns particle testing from detection into investigation.
11) Best Practices for Subvisible Particle Investigations (QC-Friendly)
When subvisible particles increase or appear unexpectedly, the goal is not only to quantify the event, but to characterize and interpret it.
Here are best practices that improve investigation outcomes:
1) Use orthogonal confirmation
When LO results are unexpected or ambiguous, confirm with an imaging-based method. Many populations that appear similar in LO data become easy to interpret once you can see them.
2) Minimize time-dependent sample changes
Subvisible particle samples may change quickly:
-
heavier particles can settle
-
silicone droplets can float and coalesce
-
dispersion can drift over time
Consistent sample handling and timely analysis improve repeatability.
3) Preserve traceability
Strong investigations include:
-
representative images
-
morphology metrics
-
clear reporting outputs
This strengthens defensibility.
4) Differentiate particle types early
A key investigation milestone is determining whether the population is:
-
droplets vs aggregates
-
fibers vs fragments
-
mixed or uniform
This quickly narrows likely sources and next steps.
5) Build a particle image library
A particle library becomes a powerful tool for:
-
trending and comparison
-
faster investigation triage
-
training and internal consistency
12) Practical Path Forward for Biopharma Teams
The most effective subvisible particle programs typically follow a practical approach:
-
use compendial LO for routine trending and compliance
-
use imaging methods for characterization and investigations
-
apply orthogonal methods to strengthen evidence and decision-making
As biologics become more complex and regulatory expectations evolve, the ability to characterize particles—visually and morphologically—becomes a key capability for modern QC.
Conclusion
Subvisible particle analysis is no longer only about size and count. In many cases, QC teams need to understand what particles look like, whether populations are mixed, and what they may indicate.
That’s why orthogonal characterization—supported by frameworks like USP <1788> and USP <1788.3>—is increasingly important in modern biologics quality programs.
The best approach is practical: maintain compendial LO testing, and add imaging-based characterization when you need visual evidence, differentiation, and stronger investigations.
If you would like to see how imaging-based particle characterization supports subvisible particle investigations, we can demonstrate analysis using:
-
protein aggregate controls
-
silicone oil droplets
-
or your own samples
Request a consultation or schedule a live demo
Reach out here: Contact Vision Analytical
FAQ
What are subvisible particles in biologics?
Subvisible particles are particles not visible to the unaided eye that may include protein aggregates, silicone oil droplets, fibers, or fragments. They are important because they may indicate formulation instability, process issues, or container closure interactions.
What is the difference between light obscuration and flow imaging microscopy?
Light obscuration reports size and count but does not provide images. Flow imaging microscopy captures images and morphology metrics, enabling particle differentiation and stronger investigation support.
What does USP <1788.3> add to particle testing guidance?
USP <1788.3> recognizes imaging-based methods as orthogonal techniques for subvisible particle characterization, supporting visual confirmation and morphology assessment.